cyclopropanecarboxylic acid, 1-hydroxy-, methyl ester
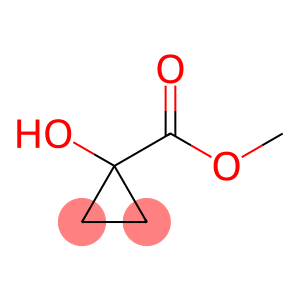
Methyl 1-hydroxycyclopropanecarboxylate
CAS: 33689-29-1
Molecular Formula: C5H8O3
cyclopropanecarboxylic acid, 1-hydroxy-, methyl ester - Names and Identifiers
cyclopropanecarboxylic acid, 1-hydroxy-, methyl ester - Physico-chemical Properties
| Molecular Formula | C5H8O3 |
| Molar Mass | 116.12 |
| Density | 1.346±0.06 g/cm3(Predicted) |
| Boling Point | 70°C/13mmHg |
| Flash Point | 17℃ |
| Solubility | Dichloromethane, Methanol |
| Vapor Presure | 3.47mmHg at 25°C |
| Appearance | Liquid |
| Color | Clear yellow |
| pKa | 13.14±0.20(Predicted) |
| Storage Condition | Sealed in dry,2-8°C |
| Refractive Index | 1.521 |
cyclopropanecarboxylic acid, 1-hydroxy-, methyl ester - Risk and Safety
| Risk Codes | 11 - Highly Flammable |
| Safety Description | 16 - Keep away from sources of ignition. |
| UN IDs | 3277 |
| HS Code | 29162090 |
cyclopropanecarboxylic acid, 1-hydroxy-, methyl ester - Reference Information
| Introduction | Methyl 1-hydroxy-1-cycloprocarboxylate is an organic intermediate. Because many compounds containing cyclopropane substructural units have good biological activity, cyclopropane has a very wide range of applications in drug design. |
| Uses | Methyl 1-hydroxy-1-cycloprocarboxylate is a carboxylate organic substance and can be used as an intermediate in organic synthesis. |
| preparation | 1-aminocyclopropyl methyl ester (3.41g,29.6mmol,1.0eq) is dissolved in 40ml of sulfuric acid aqueous solution a (1.62mL of 98% concentrated sulfuric acid is added to water to prepare, the molar equivalent of sulfuric acid is 1.0eq), the ice bath is cooled to 0-5 ℃, and 10ml of sodium nitrite (2.25g,32.6mmol,1.1eq) aqueous solution, stirring at room temperature for 1 hour. Then the above reaction droplets are added to the refluxed 100ml sulfuric acid aqueous solution B (obtained by adding 1.62mL 98% concentrated sulfuric acid into water, and the molar equivalent of sulfuric acid is 1.0eq). After dropping, stop heating and cool to room temperature. After TLC detection reaction is completed, ethyl acetate (3*100mL) is extracted three times, the organic phase is combined, dried with anhydrous magnesium sulfate, filtered and concentrated the organic phase to obtain methyl 1-hydroxy -1-cycloprocarboxylate (2.57g,22.1mmol), colorless oily liquid, and the yield is 74.8%. |
Last Update:2024-04-09 21:01:54
Supplier List
Spot supply
Product Name: 1-Hydroxycyclopropanecarboxylic acid methyl ester Visit Supplier Webpage Request for quotationCAS: 33689-29-1
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Methyl 1-hydroxycyclopropanecarboxylate Visit Supplier Webpage Request for quotationCAS: 33689-29-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: METHYL 1-HYDROXY-1-CYCLOPROPANE CARBOXYLATE, 90 Request for quotation
CAS: 33689-29-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 33689-29-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 1-Hydroxycyclopropanecarboxylic Acid Methyl Ester Visit Supplier Webpage Request for quotation
CAS: 33689-29-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 33689-29-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 1-Hydroxycyclopropanecarboxylic acid methyl ester Visit Supplier Webpage Request for quotationCAS: 33689-29-1
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Methyl 1-hydroxycyclopropanecarboxylate Visit Supplier Webpage Request for quotationCAS: 33689-29-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: METHYL 1-HYDROXY-1-CYCLOPROPANE CARBOXYLATE, 90 Request for quotation
CAS: 33689-29-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 33689-29-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 1-Hydroxycyclopropanecarboxylic Acid Methyl Ester Visit Supplier Webpage Request for quotation
CAS: 33689-29-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 33689-29-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



